Inflammatory status, arterial stiffness and central hemodynamics in hypertensive patients with metabolic syndrome
- DOI
- 10.1016/j.artres.2009.09.001How to use a DOI?
- Keywords
- Arterial stiffness; Central blood pressures; Wave reflections; Metabolic syndrome; Arterial hypertension; Inflammation
- Abstract
Introduction: Inflammatory state is activated in metabolic syndrome and may explain part of the adverse prognosis of this entity. Arterial stiffness, central blood pressures and wave reflections are independent predictors of cardiovascular risk. This study investigates the relation between low-grade inflammation and arterial stiffness and central hemodynamics in patients with metabolic syndrome.
Methods: We studied 106 consecutive hypertensive patients with metabolic syndrome. Arterial stiffness was assessed by carotid-femoral (c-f) and carotid-radial (c-r) pulse wave velocity (PWV). Central blood pressures were evaluated by pulse wave analysis and heart rate corrected augmentation index (AIx75) was used as a measure of wave reflections. White blood cell count (WBC), high sensitivity C-reactive protein (hsCRP) and fibrinogen were measured as inflammatory markers.
Results: In univariable analysis, PWVc-f correlated with both hsCRP (p<0.01) and fibrinogen (p<0.01), while PWVc-r correlated with hsCRP (p=0.05). Regarding central blood pressures, aortic SBP correlated positively with hsCRP (p<0.05) and marginally with fibrinogen (p=0.06) and WBC (p=0.08). Interestingly, no correlation was found between AIx75 and any of these biomarkers. After adjustment for age, gender, smoking, mean arterial pressure, heart rate, waist circumference, glucose, total and HDL cholesterol, PWVc-f was independently related to hsCRP (p<0.001) and fibrinogen (p<0.01), while a marginal independent correlation was also found between PWVc-r and hsCRP (p=0.06). Furthermore, aortic PP independently associated with fibrinogen (p<0.05) whereas marginal associations demonstrated between aortic SBP and hsCRP and fibrinogen (p=0.06 for both).
Conclusion: Inflammatory status is related to arterial stiffness and central blood pressures (but not to augmentation index) in hypertensives with metabolic syndrome. These findings may have implications for increased cardiovascular risk in these patients.
- Copyright
- © 2009 Association for Research into Arterial Structure and Physiology. Published by Elsevier B.V.All rights reserved.
- Open Access
- This is an open access article distributed under the CC BY-NC license.
Introduction
Metabolic syndrome (MetS) is a cluster of metabolic disorders associated with a high incidence of diabetes mellitus type II and cardiovascular events.1 Particularly for hypertension, studies have shown that hypertensives with MetS have almost two-fold greater risk of cardiovascular events compared to hypertensives without MetS and this risk increases as the number of the MetS components increases.2 Furthermore, MetS is related to inflammation, abnormal haemostasis and impaired endothelial function.3,4
Arterial stiffness, central blood pressures and wave reflections are independent predictors of cardiovascular risk.5,6 Both in the general population.7–11 and in hypertensive patients,12–14 arterial stiffness has been associated with MetS. However, little evidence exists regarding the association between central blood pressures and wave reflection indices in MetS.9,15,16 While large artery stiffness is associated with low-grade inflammation,17–20 the association of the latter with central pressures and wave reflections indices is controversial.17–19 Accordingly, in the present study we sought to investigate associations between large artery stiffness, central pressures and wave reflection indices and inflammatory markers and mediators in the setting of hypertension and MetS, two conditions particularly associated with activated inflammatory state. For this purpose, we assessed and explored relationships between pulse wave velocity (a direct measure of arterial stiffness), aortic pressures, augmentation index (an index of wave reflections), white blood cell count (WBC), high sensitivity C-reactive protein (hsCRP) and fibrinogen in never treated hypertensive patients with MetS.
Methods
The study participants were 106 non-diabetic patients with never treated essential hypertension and metabolic syndrome, recruited from the Hypertension and Peripheral Vessels Units of our Department. Office blood pressure (BP) was measured by a mercury sphygmomanometer, taking at least three measurements spaced by 1min, after having allowed the patients to rest for 10min before the examination. According to the guidelines of the European Society of Hypertension, measurements of SBP ≥140mmHg and DBP ≥90mmHg were considered as systolic and diastolic arterial hypertension, respectively. Pulse Pressure (PP) was calculated as SBP minus DBP, while mean arterial pressure (MAP) was calculated as DBP plus 1/3 PP. All measurements were conducted in the morning between 9 and 11 am, with the individual in a supine position, in a quiet environment. Participants were requested to abstain from tobacco, coffee and food at least 5h and from alcohol 12h before the examination.
Metabolic syndrome was defined according to the criteria of National Cholesterol Education Program-Third Adult Treatment Panel III 2001 (NCEP-ATP III) as the presence of arterial hypertension and two or more of the following: waist circumference >102cm (males), >88cm (females), triglycerides ≥1.7mmol/L, high-density lipoprotein (HDL) <1.03mmol/L (males), <1.30mmol/L (females) and fasting plasma glucose ≥6.1mmol/L. Patients with coronary artery disease, cerebrovascular events, peripheral artery disease and systemic inflammatory disease were excluded from the study. Use of anti-inflammatory agents (including aspirin and corticosteroids), anticoagulants and statins were also exclusion criteria for the study. Weight and height were measured in all subjects and body mass index (BMI) was calculated. Waist and hip circumference was also measured and the waist to hip ratio was calculated. The smoking status was assessed by recording the habit and duration of smoking (pack-years).
All participants gave their informed consent to participate in the study, which was approved by the local Ethics Committee.
Laboratory measurements
Blood samples were collected between 8 and 9 am with individuals in the supine position after an overnight fast, centrifuged at 3000g for 30min at 3°C and stored at −70°. Both hsCRP and fibrinogen levels were measured by immunonephelometry (Dade Behring, Marburg, Germany). The intra- and inter-assay coefficient of variation (CV) for hsCRP was <4.4% and <5.7% respectively whereas for fibrinogen was 2.7 and 2.6% respectively. White blood cell count and biochemical variables such as triglycerides, total cholesterol, HDL and plasma glucose levels were measured using standard techniques.
Arterial stiffness and wave reflections evaluation
Carotid-femoral (PWVc-f) pulse wave velocity, an established index of aortic stiffness5 and carotid-radial (PWVc-r) pulse wave velocity, a measure of medium-sized (muscular) artery stiffness, were obtained using a validated non-invasive device (Complior®, Artech Medical, Pantin, France), which allows online pulse wave recording and automatic calculation of pulse wave velocity. Three different pressure waveforms were obtained simultaneously at three sites: the right carotid, radial and femoral artery. The time delay (t) between the feet of the recorded proximal and distal waves was automatically calculated. For carotid-femoral segment, the distance (d) was estimated by subtracting the distance from carotid location to the sternal notch from the distance between the sternal notch and the femoral site. For carotid-radial segment, the distance was estimated by subtracting the distance from carotid location to the sternal notch from the distance between the carotid location and the radial site of measurement. The mean PWV of at least 10 consecutive pressure waveforms was calculated for further analysis.
Central blood pressures [aortic systolic blood pressure (aortic SBP) and aortic pulse pressure, (aortic PP)] and augmentation index (AIx) were estimated by pulse wave analysis. The peripheral pulse wave was recorded from the radial artery using the method of applanation tonometry. The radial pulse wave was transformed into the central pulse wave of the aorta through analysis of pulse wave with the Sphygmocor device (Sphygmocor, AtCor Medical, Sydney, Australia).5,6 Augmentation Index was measured as an index of wave reflections. AIx is a composite measure of the magnitude of wave reflection and arterial stiffness which affects timing of wave reflections. Augmented Pressure (AP) is the pressure added to the incident wave by the returning reflected wave and represents the increased afterload that left ventricle must cope with. AIx is defined as AP divided by central pulse pressure and is expressed as a percentage. Measurements were performed by a single experienced investigator. AIx was averaged from ten to twelve successive waves and it was corrected for a steady heart rate of 75beats/min (AIx75).
Statistical analysis
Between groups, Student t-test for unpaired measures or the chi-square test were used to compare continuous and categorical variables respectively. Correlations between variables were evaluated by calculation of the Pearson correlation coefficient. hsCRP was log transformed due to its skewed distribution. We applied linear regression models to evaluate the association between the investigated arterial stiffness indices (PWVc-f and PWVc-r as dependent variables), as well as between aortic SBP and aortic PP, and loghsCRP, WBC and fibrinogen (as independent variables) after adjusting for age, gender, smoking, MAP (it was used as an independent variable only in PWV models), heart rate, glucose, total cholesterol, HDL cholesterol and waist circumference. The assumptions for linearity and homoscedasity were tested based on the standardized residuals plots, while the assumption of normality for the dependent variable was tested using the Kolmogorov-Smirnov criterion. Exact p values <0.05 were considered statistically significant. Data analysis was performed with SPSS software, version 13.0 (SPSS Inc., Chicago, Illinois, USA).
Results
The baseline characteristics of the study population are shown in Table 1.
In univariable analysis, PWVc-f correlated with age (r=0.48, p<0.001), peripheral SBP (r=0.46, p<0.001), aortic SBP and aortic PP (r=0.40, p<0.001 for both), hsCRP (r=0.33, p<0.01) and fibrinogen (r=0.30, p<0.01). A significant association was found between PWVc-f and PWVc-r (r=0.19, p<0.05), as well as between PWVc-f and AIx75 (r=0.25, p=0.01). PWVc-f did not differ between smokers and non smokers (7.99±1.36 vs 8.35±1.32m/s, p=NS) or between males and females (8.14±1.45 vs 8.41±1.04m/s, p=NS).
PWVc-r showed a positive correlation with peripheral SBP (r=0.26, p<0.01), peripheral DBP (r=0.26, p<0.01) and aortic SBP (r=0.29, p<0.01) while an inverse relationship was observed with HDL (r=−0.22, p<0.05). Furthermore, PWVc-r was related to hsCRP (r=0.19, p=0.05) but no association was found between PWVc-r and fibrinogen (p=NS). No difference was shown in PWVc-r values between smokers and non smokers (8.79±1.15 vs 8.57±1.11m/s, p=NS), whereas males had increased PWVc-r compared to females (8.82±1.16 vs 8.34±1.01m/s, p<0.05).
AIx75 was associated with age (r=0.48, p<0.001), peripheral SBP (r=0.25, p<0.05), peripheral DBP (r=0.19, p<0.05), aortic SBP (r=0.45, p<0.001), aortic PP (r=0.40, p<0.001) and glucose level (r=0.22, p<0.05). Interestingly, a positive association was observed with HDL (r=0.23, p<0.05) and an inverse with waist/hip ratio (r=−0.29, p<0.01). No association was found between AIx75 and any of the measured inflammatory markers (p=NS for all).
Aortic SBP was related to hsCRP (r=0.21, p<0.05) and marginally to fibrinogen (r=0.18, p=0.06) and WBC (r=0.17, p=0.08). Aortic PP showed a positive correlation with age (r=0.31, p<0.01) and fibrinogen (r=0.20, p<0.05).
Regarding the inflammatory parameters, hsCRP correlated with peripheral SBP (r=0.24, p=0.01), peripheral DBP (r=0.25, p<0.05) and fibrinogen (r=0.38, p<0.001). Moreover, fibrinogen levels were higher in smokers compared to non smokers (369.59±85.82 vs 333.67±76.35mg/dl, p<0.05).
After linear regression analysis, an independent association was demonstrated between PWVc-f and both hsCRP (Table 2, Fig. 1) and fibrinogen (Table 3, Fig. 1). A marginal correlation between PWVc-r and hsCRP (Table 2) was also found. Regarding central blood pressures, aortic SBP was marginally associated with hsCRP (Table 2) and fibrinogen (Table 3), while a significant independent correlation was found between aortic PP and fibrinogen (Table 3, Fig. 2). No independent relationships emerged between AIx75 and any of the inflammatory biomarkers.
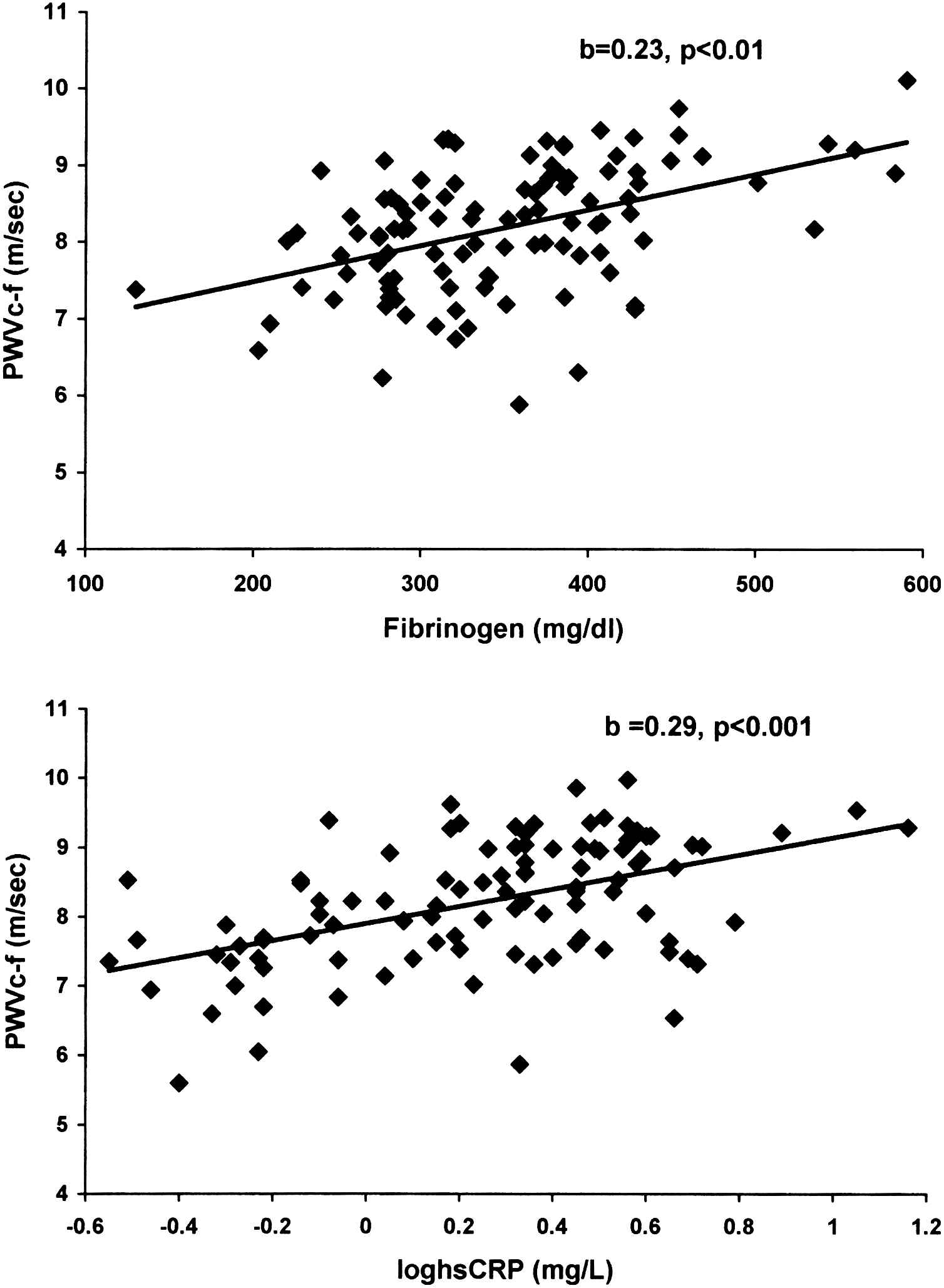
Independent relationship of carotid-femoral pulse wave velocity (PWVc-f) with fibrinogen (upper panel) and high sensitivity C-reactive protein[hsCRP] (lower panel) in hypertensive patients with metabolic syndrome.
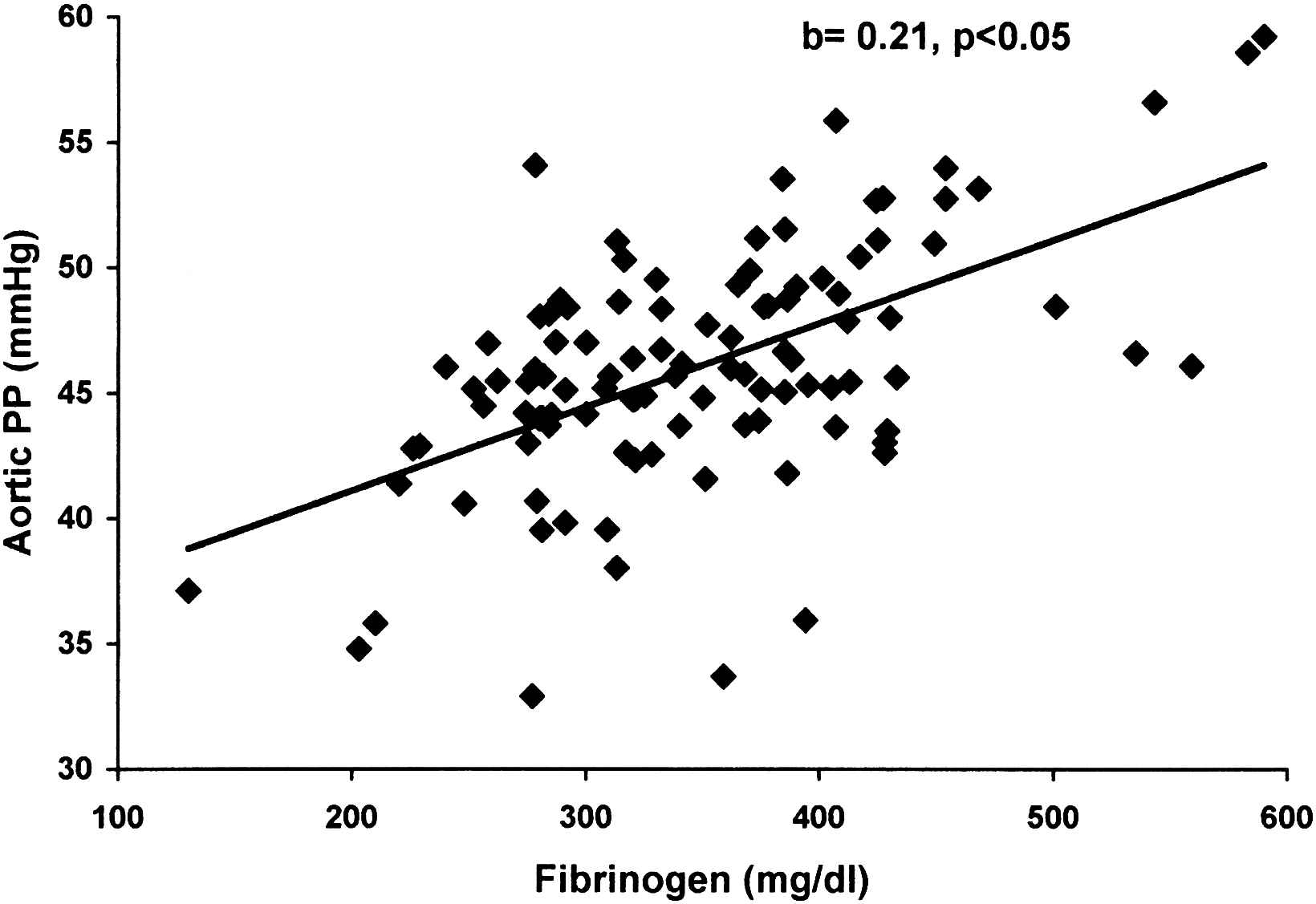
Independent association of aortic pulse pressure (aortic PP) with fibrinogen in hypertensive patients with metabolic syndrome.
| Parameter | Value |
|---|---|
| Age (years) | 50.3±9.8 |
| Males (%) | 68.0 |
| Smokers (%) | 41.5 |
| BMI (kg/m2) | 30.8±3.9 |
| Waist/Hip ratio | 0.93±0.07 |
| Total cholesterol (mmol/L) | 5.79±1.11 |
| Triglycerides (mmol/L) | 2.01±1.05 |
| HDL (mmol/L) | 1.12±0.24 |
| Glucose (mmol/L) | 5.68±0.85 |
| hsCRP (mg/L) | 2.44±2.08 |
| Total fibrinogen (mg/dl) | 348±82 |
| Peripheral SBP (mmHg) | 152±18 |
| Peripheral DBP (mmHg) | 91±10 |
| Peripheral MAP (mmHg) | 112±12 |
| Peripheral PP (mmHg) | 59±15 |
| Heart rate (beats/min) | 71±9 |
| Aortic SBP (mmHg) | 140±17 |
| Aortic DBP (mmHg) | 92±10 |
| Aortic PP (mmHg) | 46±13 |
| PWVc-f (m/s) | 8.21±1.34 |
| PWVc-r (m/s) | 8.66±1.13 |
| AIx75 (%) | 25.6±9.0 |
Continuous variables are presented as mean value ±1 standard deviation.
Abbreviations: BMI, body mass index; HDL, high-density lipoprotein; hsCRP, high sensitivity C-reactive protein; SBP, systolic blood pressure; DBP, diastolic blood pressure; MAP, mean arterial pressure; PWVc-f, carotid-femoral pulse wave velocity; PWVc-r, carotid-radial pulse wave velocity; AIx75, heart rate corrected augmentation index.
Baseline characteristics of the study population (N=106).
| Standardized b coefficient | p value | |
|---|---|---|
| PWVc-f | ||
| Age | 0.55 | <0.001 |
| MAP | 0.31 | <0.001 |
| loghsCRP | 0.29 | <0.001 |
| Adjusted R2=0.46 | ||
| PWVc-r | ||
| Male gender | 0.24 | <0.05 |
| Waist | −0.19 | 0.07 |
| MAP | 0.27 | <0.01 |
| loghsCRP | 0.18 | 0.06 |
| Adjusted R2=0.18 | ||
| Aortic SBP | ||
| loghsCRP | 0.18 | 0.06 |
| Adjusted R2=0.08 | ||
Abbreviations: PWVc-f, carotid-femoral pulse wave velocity; PWVc-r, carotid-radial pulse wave velocity; SBP, systolic blood pressure; hsCRP, high sensitivity C-reactive protein; MAP, mean arterial pressure.
Association of PWVc-f, PWVc-r and aortic SBP with hsCRP after adjusting for several confounders.
| Standardized b coefficient | p value | |
|---|---|---|
| PWVc-f | ||
| Age | 0.47 | <0.001 |
| MAP | 0.35 | <0.001 |
| Fibrinogen | 0.23 | <0.01 |
| Adjusted R2=0.43 | ||
| Aortic SBP | ||
| Fibrinogen | 0.18 | 0.06 |
| Adjusted R2=0.07 | ||
| Aortic PP | ||
| Age | 0.28 | <0.01 |
| Fibrinogen | 0.21 | <0.05 |
| Adjusted R2=0.14 | ||
Abbreviations: PWVc-f, carotid-femoral pulse wave velocity; SBP, systolic blood pressure; PP, pulse pressure; MAP, mean arterial pressure.
Association of PWVc-f, aortic SBP and aortic PP with fibrinogen after adjusting for several confounders.
Discussion
The present study shows that arterial stiffness and central blood pressures are independently associated with inflammation in hypertensive patients with MetS. An additional novel finding of our study is that no association can be established between wave reflections indices, such as the augmentation index, and any of the inflammatory parameters in such patients. These findings extend recent studies from our Department showing that both hsCRP and fibrinogen are associated with carotid-femoral PWV (but not with AIx) in hypertensives,19 in hypertensives without the metabolic syndrome (unpublished data), and in healthy individuals.21 Furthermore, they are in line with studies showing that in general population with MetS, ankle-brachial PWV is related to hsCR,22 and that in young women with MetS, hsCRP and fibrinogen explained part of the association of PWV with MetS.23 Although no control group was included in the present study, our previous data on hypertensive patients and healthy controls showed similarities regarding the positive association of PWV with hsCRP and the absence of relationship between AIx and hsCRP.19
Arterial stiffness and central hemodynamics in metabolic syndrome: The role of inflammation
Arterial stiffness and inflammation in MetS
Central obesity has been associated with increased arterial stiffness24 and thus it may represent a potential link between increased arterial stiffness and inflammation in MetS. Indeed, through the secretion of IL-6, adipose tissue regulates the production of hsCRP from hepatocytes.25 In turn, hsCRP upregulates angiotensin type I receptor,26 stimulates the migration and proliferation of smooth muscle cells, induces the production of reactive oxygen species and decreases NO bioavailability (although the latter is controversial).27,28 Furthermore, adipocytokines (cytokines that are released from adipose tissue) have also been associated with arterial stiffness. In hypertensive population, low levels of adiponectin in conjunction with high levels of hsCRP exert an additive, detrimental effect on aortic stiffness.29 However, as regards possible mediators, it should be noted that recent data on the polymorphisms of CRP support the hypothesis of an inverse causal relationship or residual confounding between CRP and arterial stiffness,30,31 while in our acute experiments, there was a temporal dissociation between the changes of hsCRP and arterial stiffness.17
Regarding fibrinogen, whether it is merely an indicator of the level of inflammation or it affects arterial stiffness by itself, is difficult to clarify. Fibrinogen is an acute phase reactant protein which, similarly to hsCRP, is regulated by proinflammatory cytokines,25 and thus it may be indirectly related to arterial stiffness. On the other hand, fibrinogen may accelerate the inflammatory process by upregulating the expression of inflammatory chemokines.32 Furthermore, considering that endothelium regulates arterial stiffness33 fibrinogen may affect arterial stiffness by promoting endothelial dysfunction.34,35 Finally, fibrinogen may migrate into the arterial intima and form fibrin and microthrombi, which in turn activate the inflammatory cascade, and impair normal arterial function.36
Central hemodynamics and inflammation in MetS
Despite the significant association of PWV with inflammatory status, central pressures and AIx were not uniformly associated with inflammatory indices in our study. Central pressures and wave reflection indices depend both on the timing of the merging of the incident and reflected wave (which is in turn dependent on pulse wave velocity) and on the magnitude of the reflected wave (which is dependent on vascular tone). Since MetS is an inflammatory disease, a degree of peripheral vasodilatation induced by inflammation might decrease the magnitude of reflected waves. Conversely, during chronic inflammation, higher pulse wave velocity (resulting from increased arterial stiffness) tends to shift earlier the merging point and thus increase central pressures and wave reflection indices. Accordingly, our findings may imply that each component may affect differently central pressures and AIx: relative preponderance of arterial stiffness in the determination of central pressure leads to positive associations with inflammation, whereas relative preponderance of vasodilation in the determination of AIx leads to absence of association. Our present finding regarding AIx is further corroborated by previous studies from our and other groups showing inverse17 or no relationship19 of AIx with inflammatory markers and absence of a relationship between AIx and MetS.9,15,16
Central obesity (as indicated by waist circumference) might also explain the absence of relationship between inflammatory factors and wave reflections in our study. Indeed, although there is a consistent positive correlation between aortic stiffness and central obesity, results regarding the associations of central obesity with wave reflections are both positive37 and negative.38
Clinical implications
The findings of the present study may have important clinical implications. Although no etiological associations can be established, it can be inferred that a part of the risk in hypertensives with MetS is mediated through increased arterial stiffness and central pressures given their pathophysiological and prognostic role.5,39,40 A stiff aorta increases left ventricular afterload, induces left ventricular hypertrophy and disturbs diastolic coronary perfusion, thus unbalancing coronary perfusion/myocardial demand equilibrium.41 Accordingly, targeting arterial stiffness through modulation of the inflammatory state could possibly improve prognosis. Concerning pharmacological treatment, angiotensin converting enzyme (ACE) inhiditors and angiotensin II receptor antagonists have been shown to favourably affect both arterial stiffness and inflammatory status.42–45 Furthermore, ACE inhibitors46 and nebivolol47 have been found to decrease fibrinogen levels in hypertensive patients. Statins48 and aspirin49 may be particularly favourable in patients with MetS due to their anti-inflammatory effects. Beyond pharmacological treatment, physical activity, specific diets, such as the Mediterranean, and weight loss may also lead to reduction of arterial stiffness through their effect on inflammation.50,51
Conclusion
In conclusion, the activated inflammatory state in hypertensive patients with the metabolic syndrome is independently associated with arterial stiffness and central blood pressures. Accordingly, part of the increased cardiovascular risk of metabolic syndrome may be conveyed through interrelationships between arterial stiffness/central blood pressures and inflammation. However, no etiological relationships can be established from the present study and further studies are warranted to establish possible causal effects and to explore the effect of anti-inflammatory interventions.
Conflict of interest
None
Funding
None
References
Cite this article
TY - JOUR AU - Panagiota Pietri AU - Charalambos Vlachopoulos AU - Konstantinos Aznaouridis AU - Katerina Baou AU - Panagiotis Xaplanteris AU - Ioanna Dima AU - Gregory Vyssoulis AU - Christodoulos Stefanadis PY - 2009 DA - 2009/10/28 TI - Inflammatory status, arterial stiffness and central hemodynamics in hypertensive patients with metabolic syndrome JO - Artery Research SP - 115 EP - 121 VL - 3 IS - 3 SN - 1876-4401 UR - https://doi.org/10.1016/j.artres.2009.09.001 DO - 10.1016/j.artres.2009.09.001 ID - Pietri2009 ER -
