Prevalence of zoonotic tuberculosis and associated risk factors in Central Indian populations
- DOI
- 10.1016/j.jegh.2017.08.007How to use a DOI?
- Keywords
- Duplex PCR; Mycobacterium bovis; Tuberculosis; Zoonosis
- Abstract
In the present study, we aimed to estimate the occurrence of bovine tuberculosis (TB) and examine the determinants of distribution of the disease in three high-risk populations of Central India. A prospective cohort study was conducted in Central India between March 2014 and June 2015. Based on the requisite inclusion criteria, we recruited a total of 301 participants whose blood samples were subjected to polymerase chain reaction-based detection and differentiation of Mycobacterium bovis and Mycobacterium tuberculosis. M. bovis was detected in 11.4%, 8.9%, and 12.6% of the recruited participants belonging to three distinct population groups (Groups A, B, and C, respectively). The highest proportion of cases infected with M. bovis was observed in Group C, who lived in the high TB endemic region. Previous contact with active TB cases (odds ratio = 3.7; 95% confidence interval, 0.9612–14.4533) and raw milk consumption (odds ratio = 5.3472; 95% confidence interval, 1.9590–14.5956) were found to be important determinants of bovine TB in this population. The high incidence rates of bovine TB in the Central Indian populations indicate the substantial consequences of this disease for some population groups and settings. However, more research is necessary to identify the main transmission drivers in these areas.
- Copyright
- © 2017 Ministry of Health, Saudi Arabia. Published by Elsevier Ltd.
- Open Access
- This is an open access article under the CC BY-NC-ND license (http://creativecommons.org/licenses/by-nc-nd/4.0/).
1. Introduction
Tuberculosis (TB) remains the foremost leading cause of death worldwide, affecting more than 9 million people every year. Although Mycobacterium tuberculosis is the most common cause of human TB, unknown proportions of TB cases are considered to be attributable to Mycobacterium bovis infection, which is also termed bovine TB [1]. The infection currently poses a major concern in human populations in developing countries, as humans and animals share the same microenvironment. It has been estimated that zoonotic transmission of M. bovis is responsible for 10–15% of new human TB cases in developing countries [2]. The disease in humans primarily occurs through close contact with infected cattle or consumption of improperly cooked beef and drinking of unpasteurized milk and milk products [3,4]. People in specific occupations such as veterinarians, farmers, and abattoir workers are considered to be more at risk [5].
The prevalence of bovine TB infection and associated risk factors have been insufficiently characterized in the Indian population that is considerably dependent on agriculture for its livelihood. It has been suggested that poverty, poor dietary habits, close physical contact between humans and animals, and inadequate disease control measures facilitate the transmission of zoonoses [6]; however, there is no substantial evidence in the Indian context to justify the hypothesis.
Currently available tests used for identification of M. bovis are based on bacterial isolation and biochemical tests, which are both time-consuming with low diagnostic accuracy [7]. Furthermore, TB caused by M. tuberculosis in humans is clinically and radiologically identical to TB caused by M. bovis [8]. These problems were overcome by molecular techniques to some extent; however, this technique could not identify the mycobacterial pathogens to the species level. In the present study, with the purpose of detecting zoonotic transmission of M. bovis and simultaneous differentiation of members of M. tuberculosis complex species (MTBC), we targeted the regions of difference (RDs) through a duplex polymerase chain reaction (PCR) assay.
As there are limited reports on the occurrence of bovine TB in India, we aimed to estimate its incidence among participants belonging to three distinct population groups and settings based on assessment of risk factors and occupational exposure to animals. The participants for this study were recruited from Achalpur, Amravati, Pilkapar, and Nagpur, located in the Central Indian region of Maharashtra, India. A comparative analytical study was then carried out to evaluate the determining factors that may influence the distribution of the disease in each population.
2. Material and methods
2.1. Ethics statement
The study was approved by the Ethical Committee of Central India Institute of Medical Sciences, Nagpur. All clinical investigations were conducted according to the principles expressed in the declaration of Helsinki 1975, as revised in 1983. Written consent forms were obtained from all recruited participants after they were given a detailed oral explanation of the study.
2.2. Study design and participants
A total of 433 participants were enrolled from three different populations within Central India region through camps organized between March 2014 and June 2015. These population groups were approached with the help of a local health care practitioner. A survey using a standardized questionnaire investigating exposure to TB cases; consumption of meat, unpasteurized milk, and milk products; as well as other sociodemographic parameters such as age, sex, occupation, duration of exposure to animals, and type of animal reared along with clinical history was conducted (Table S1). Active TB was investigated in individuals with respiratory or systemic symptoms by acid fast bacilli (AFB) smear and culture of appropriate sputum samples and chest X-ray. The details of the recruited populations are described in the following subsections.
2.2.1. Group A: Farmers, dairy workers, and livestock keepers
Participants in this group included farmers from Achalpur and Pilkapar villages of the state of Maharashtra in Central India. Most of the population had agriculture as their major occupation. Some of them were also involved in dairy production and livestock keeping. The farmers or members of their family were personally involved in the maintenance of these animals. The animals reared included Indian breed of ox, cows, buffaloes, and calves. Milking and delivery of pregnant animals were also done by the members of the household.
2.2.2. Group B: Zookeepers and animal handlers
This population included zookeepers and veterinarians from a specific locality within the Nagpur district. The participants from this group were involved in activities such as guarding, cleaning, feeding, and maintenance of animals in the zoo. The veterinarians recruited in this group were involved in routine health checkup, semen analysis, treatment, and post mortem of deceased animals.
2.2.3. Group C: Residents of high TB endemic area
This population belonged to a specific locality of Nagpur district, in the Vidarbha region of Maharashtra. The majority of the population routinely included meat and other animal products in their diet. The endemic area had high crowding index with an average of six to eight individuals living in small poorly ventilated rooms. The majority of the population had poor socioeconomic status and living conditions characterized by lack of sanitation and poor hygiene. Some households also participated in cattle and goat rearing.
Of the 433 participants enrolled in this study, 84 participants were excluded based on their refusal to give blood. The remaining 349 participants who matched the inclusion criteria were selected for the study. Among these, pregnant women (n = 8), children below the age of 10 years (n = 18), and individuals with fungal or viral infections (n = 22) were also excluded from the study. Fig. 1 represents the inclusion/exclusion criteria adopted for recruitment of the study populations.
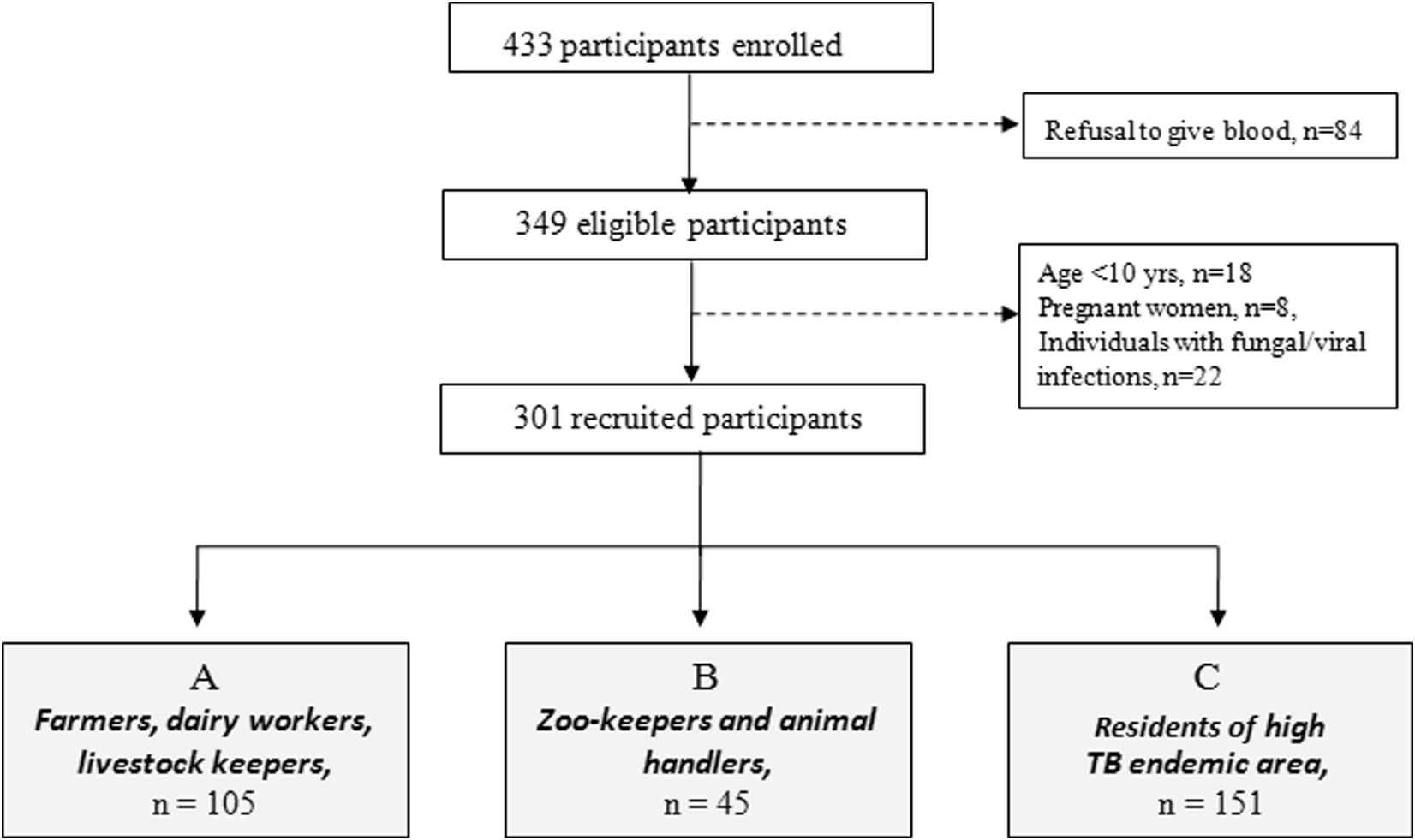
Inclusion/exclusion criteria adopted for recruitment of the study population. The population was categorized into three groups based on their occupation and origin. Gray boxes indicate the groups included in the final analysis.
2.3. Sample collection
For DNA isolation, 2.5 mL blood was collected in 5-mL sterile syringe BD and dispensed in a sterile vacutainer with coagulant EDTA). Each sample was labeled with a code that corresponded to the study location and identification of the individual.
2.4. DNA isolation and quantification
DNA was extracted from blood samples using the phenol chloroform extraction method described by Deshpande et al. [9], in which 6 mL phosphate buffered saline (PBS) was added to 2 mL blood and mixed thoroughly. Next, 8 mL of blood + PBS was added slowly from the sides of the tube to 4 mL Histopaque and centrifuged at 2000 rpm for 10 min. The buffy coat was transferred to another tube, and an equal volume of PBS was added and the tube was centrifuged again at 2000 rpm for 10 min. The supernatant was discarded and the pellet was suspended in 500 μL PBS, 15 μL 10% SDS, and 3 μL proteinase K (20 mg/ml), then mixed and incubated at 55 °C for 1 and ½ hour. After incubation, 100 μL of 5 M NaCl and 80 μL of high-salt cetyl-trimethyl-ammonium bromide (CTAB) (containing 4 M NaCl, 1.8% CTAB) was added and mixed followed by incubation at 65 °C for 10 min. An approximately equal volume (350 μL) of phenol and of chloroform/isoamyl alcohol (24:1) was added, mixed thoroughly, and centrifuged for 10 min in a microcentrifuge at 12,000 rpm. The aqueous viscous supernatant was carefully decanted and transferred to a new tube. An equal volume of phenol/chloroform-isoamyl alcohol (1:1) was added, followed by a 10-min spin at 12,000 rpm. The aqueous layer was separated and then mixed with 30 μl of 3 M sodium acetate and 0.6 vol of isopropanol to obtain a precipitate. The precipitated nucleic acids were washed with 70% ethanol, dried and resuspended in 30 μL Tris–EDTA (TE) buffer, and then stored at −20 °C prior to use. DNA concentrations for all samples and strains used in this study were determined with the Quant-iT dsDNA HS assay kit using a Qubit fluorometer (Invitrogen).
2.5. Duplex PCR
For determination to the species level of the mycobacterial pathogens—namely, M. tuberculosis, M. bovis, and M. bovis bacille Calmette–Guerin (BCG)—two genetic regions RD4 and RD1 were amplified using a duplex approach. Primers used in this study are shown in Table 1. RD4 is an RD in the bovine lineage. The use of RD4 flanking primers ensured that the PCR products were formed only if the deletion was present [10]. The genes in the RD1 region belong to the esat6 gene cluster. Early secretory antigenic target-6 (ESAT-6) is a potent stimulator of the immune system, and is an antigen recognized during the early stages of infection. The RD1 region of M. tuberculosis is considered to be the primary attenuating deletion in the related vaccine strain M. bovis BCG [11].
| PCR | Primers | Sequence | Annealing temperature (°C) | Amplicon size (bb) | Reference |
|---|---|---|---|---|---|
| RD4 | F | 5′-AATGGTTTGGTCATGACGCCTTC-3′ | 58 | 176 | Taylor et al. [10] |
| R | 5′-CCCGTAGCGTTACTGAGAAATTGC-3′ | ||||
| RD1 | F | 5′-CCCTTTCTCGTGTTTATAGTTTGA-3′ | 60 | 110 | Halse et al. [11] |
| R | 5′-GCCATATCGTCCGGAGCTT-3′ |
PCR = polymerase chain reaction; RD = region of difference.
Primer sequences for RD region analysis.
The duplex PCR reactions were carried out using 10× PCR buffer, 1.5 MgCl2, 0.8 mM dNTP (deoxynucleotide triphosphate), 0.4 μM of RD1 F/R primer, and 0.2 μM of RD4 F/R primer and 1.25 U of Taq DNA polymerase. The amplification procedure consisted of initial denaturation at 95 °C for 7 min and 35 cycles each of denaturation at 95 °C for 1 min, annealing at 59 °C for 1 min, and extension at 72 °C for 1 min followed by a final extension step at 72 °C for 10 min. A positive control, DNA extracted from M. bovis (ATCC BAA-935) culture, and a negative-no template control were included in each run.
2.6. PCR minimum detection limit
The analytical sensitivity of the duplex PCR assay was determined using 10-fold serial dilutions from 107 fg to 10 fg of DNA (equivalent to 106–10° genome copies) isolated from M. tuberculosis (ATCC 25177), M. bovis (ATCC BAA-935), and M. bovis BCG Pasteur (ATCC 35734) reference strains. To assess the specificity, the concentration of the DNA templates from each reference strain was adjusted to 10 ng/µL and subjected to PCR.
2.7. Interpretation of the results of duplex PCR
The PCR amplicons were analyzed on a 2% agarose gel and stained with ethidium bromide. The amplified products were then visualized under UV light. Comparative analysis on electrophoresis of the PCR products generated by the two sets of primer pairs showed the ability to distinguish between M. tuberculosis, M. bovis, and M. bovis BCG. The duplex PCR was considered positive for M. bovis when bands of both 176 bp and 110 bp were seen; the result was considered positive for M. tuberculosis when a band of only 110 bp was seen and positive for M. bovis BCG when a band of 176 bp was visualized on the gel.
2.8. DNA sequencing analysis
The PCR products were purified and sequenced using Sanger’s dideoxy chain termination method at the SciGenom Labs (Cochin, India). Sequences were verified by Basic Local Alignment Search Tool (BLAST) search using the NCBI National Center for Biotechnology Information (NCBI) website.
2.9. Statistical analysis
The frequencies (percentage) of demographics, clinical factors, and risk factors were measured on a nominal scale. Comparison between groups was performed using the chi-square test in MedCalc statistical software (version 10.1.2.0), and a difference with p < 0.05 was considered significant. Odds ratios (ORs) along with 95% confidence intervals (CIs) were obtained for each factor using the bivariate analysis done using the SPSS software (version 22.0).
3. Results
3.1. Baseline characteristics of the populations under study
The baseline characteristics of the populations under study are represented in Table 2. A total of 179 (59.5%) males and 122 (40.5%) females were enrolled in this study. A significantly higher percentage of the population from Group A (59.1%) and Group C (55%) belonged to the 18–40 years age group. On the contrary, a higher proportion of the population from Group B (75.6%) belonged to the >40 years age group. Reported clinical symptoms at the time of the study were compared in each population. A significantly higher percentage of participants with fever (39.7%), chills (34.4%), cough with expectoration (41.7%), abdominal pain (37.7%), chest pain (20.5%), weight loss (27.1%), night sweating (25.2%), headache (55.6%), and loss of appetite (37.1%) belonged to the population from Group C (p < 0.0001). By contrast, a significant number of (p < 0.0001) participants with body ache (43.8%) belonged to the population from Group A. Risk factors such contact with active TB case (51%) and living in an endemic area (100%) were more incident in Group C (26.82%) as compared to the other groups (p < 0.0001). Exposure to animals and raw milk consumption was more common in Group B (80%) and Group A (48.6%), respectively, and the difference was statistically significant (p < 0.0001).
| Characteristics | Aa (n = 105) |
Bb (n = 45) |
Cc (n = 151) |
Chi-square | df | Significance level (p) | Contingency coefficient |
|---|---|---|---|---|---|---|---|
| Demographic characteristics | |||||||
| Age (y) | |||||||
| <18 | 2 (1.9) | 0 (0) | 12 (7.9) | 5.786 | 1 | 0.0162 | 0.541 |
| 18–40 | 62 (59.1) | 11 (24.4) | 83 (55) | 52.73 | 2 | <0.0001 | 0.503 |
| >40 | 41 (39) | 34 (75.6) | 56 (37.1) | 5.786 | 2 | 0.0554 | 0.206 |
| Sex | |||||||
| Male | 72 (68.6) | 41 (91.1) | 66 (43.7) | 9.061 | 2 | 0.0108 | 0.22 |
| Female | 33 (31.4) | 4 (8.9) | 85 (56.3) | 82.836 | 2 | <0.0001 | 0.636 |
| Clinical characteristics | |||||||
| Fever | 33 (31.4) | 3 (6.7) | 60 (39.7) | 50.813 | 2 | <0.0001 | 0.588 |
| Chills | 12 (11.4) | 3 (6.7) | 52 (34.4) | 60.925 | 2 | <0.0001 | 0.69 |
| Cough with expectoration | 41 (39) | 8 (17.8) | 63 (41.7) | 41.054 | 2 | <0.0001 | 0.518 |
| Abdominal pain | 19 (18.1) | 1 (2.2) | 57 (37.7) | 63.688 | 2 | <0.0001 | 0.673 |
| Chest pain | 17 (16.1) | 2 (4.4) | 31 (20.5) | 25.24 | 2 | <0.0001 | 0.579 |
| Weight loss | 15 (14.3) | 8 (17.8) | 41 (27.1) | 28.344 | 2 | <0.0001 | 0.554 |
| Night sweating | 13 (12.4) | 4 (8.9) | 38 (25.2) | 33.855 | 2 | <0.0001 | 0.617 |
| Headache | 33 (31.4) | 12 (26.7) | 84 (55.6) | 63.767 | 2 | <0.0001 | 0.575 |
| Body ache | 46 (43.8) | 4 (8.9) | 29 (19.2) | 33.899 | 2 | <0.0001 | 0.548 |
| Loss of appetite | 27 (25.7) | 2 (4.4) | 56 (37.1) | 51.553 | 2 | <0.0001 | 0.614 |
| Risk factors | |||||||
| Previous contact with TB case | 15 (14.3) | 3 (6.7) | 77 (51) | 97.562 | 2 | <0.0001 | 0.71 |
| Living in a high endemic area | 20 (19) | 2 (4.4) | 151 (100) | 229.399 | 2 | <0.0001 | 0.755 |
| Animal contact | 82 (78.1) | 36 (80) | 49 (32.5) | 20.204 | 2 | <0.0002 | 0.329 |
| Raw milk consumption | 51 (48.6) | 2 (4.4) | 38 (25.2) | 42.484 | 2 | <0.0001 | 0.564 |
df = degree of freedom; TB = tuberculosis.
Farmers, dairy workers and livestock keepers.
Zookeepers and animal handlers.
Residents of high TB endemic area.
Demographic and clinical characteristics of the study populations (n = 301).
3.2. Detection and differentiation of M. bovis and M. tuberculosis by duplex PCR
A total of 105 samples were collected from Group A, among which 25 samples were positive by the duplex PCR assay. Of these 25 cases, M. bovis was detected in 12 (11.4%) and M. tuberculosis was detected in 13 (12.4%) cases. Forty-five samples were collected from the participants belonging to Group B. Of these, 11 (24.4%) were found to duplex PCR positive with four (8.9%) cases and seven (15.6%) cases infected with M. bovis and M. tuberculosis respectively. A total of 151 samples collected from Group C were subjected to duplex PCR assay, of which 60 (39.7%) were found to be positive. M. bovis was identified in 19 (12.6%) cases and M. tuberculosis was identified in 41 (27.2%) cases. The detection and differentiation M. tuberculosis and M. bovis in blood samples is represented in Fig. 2. The total positivity observed by the duplex PCR assay is shown in Table 3.

Duplex polymerase chain reaction (PCR) for detecting and differentiating Mycobacterium bovis, Mycobacterium tuberculosis, and M. bovis BCG. (A) The ethidium bromide-stained amplification products of L2: M. bovis, L3: M. bovis BCG, and L4: M. tuberculosis when electrophoresed on 2% agarose gel. The 176-bp and 110-bp products obtained are indicated. (B) L1: 100 bp molecular ladder; L2: positive control; L3 and L4: samples with M. bovis infection. BCG = bacille Calmette–Guerin.
| Group | No of samples collected | Duplex PCR positivity No. (%) |
Mycobacterium bovis No. (%) |
Mycobacterium tuberculosis No. (%) |
|---|---|---|---|---|
| A | 105 | 25 (23.8%) | 12 (11.4%) | 13 (12.4) |
| B | 45 | 11 (24.4%) | 4 (8.9%) | 7 (15.6%) |
| C | 151 | 60 (39.7%) | 19 (12.6%) | 41 (27.2%) |
PCR = polymerase chain reaction.
Duplex PCR positivity in the three populations under study.
3.3. Population wise distribution of bovine TB
Fig. 3 shows the population-wise distribution of bovine TB. PCR positivity for M. bovis induced TB was found to be highest in Group C (12.6%), consisting of residents from the high TB endemic region, followed by Group A with 11.4% positivity. This group consisted of farmers, dairy workers, and livestock keepers. PCR positivity was found to be the lowest (8.9%) in Group B, which consisted of zookeepers and animal handlers.
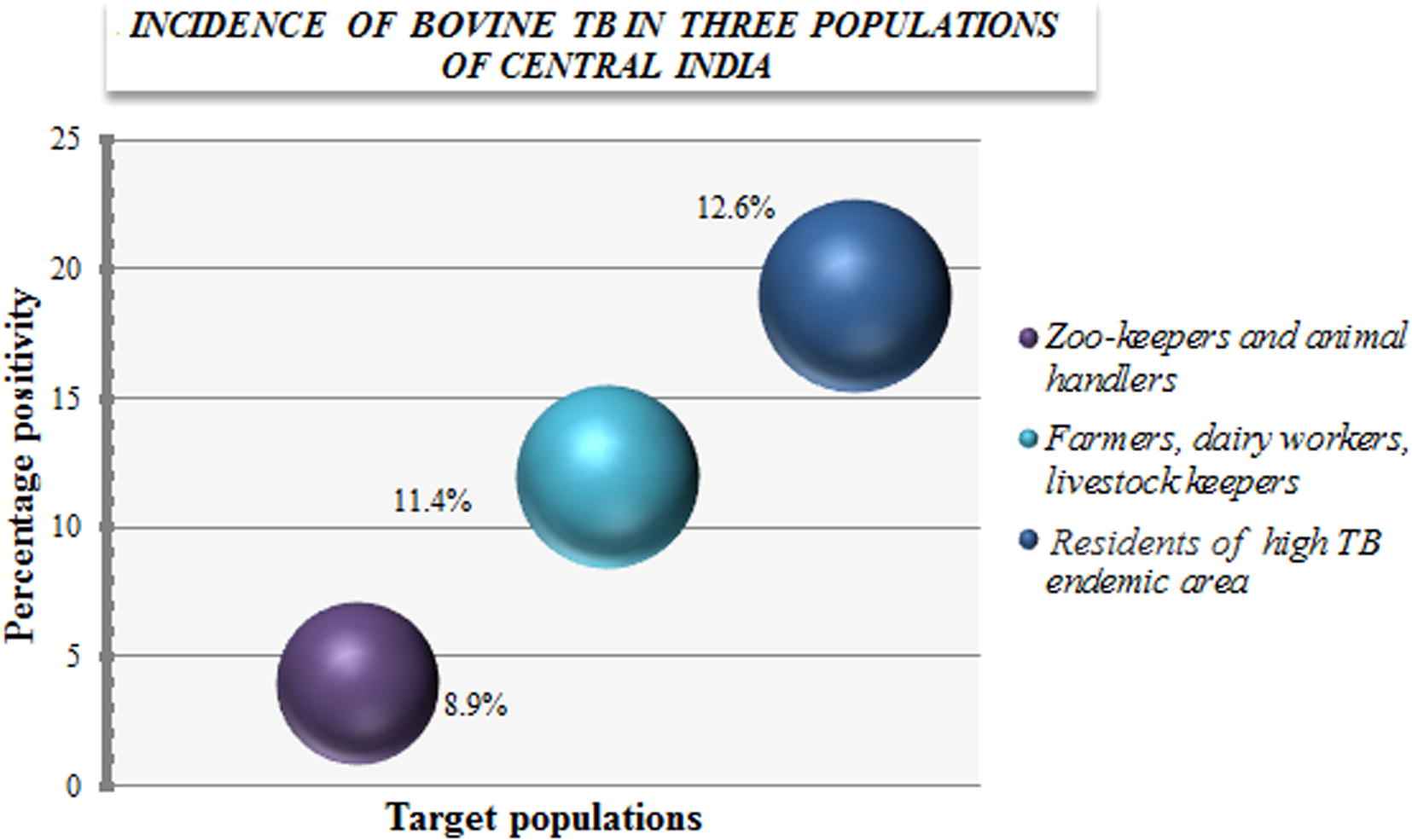
Population-wise distribution of bovine tuberculosis (TB) in Central India. Positivity (%) of Mycobacterium bovis-induced TB infection among three population groups.
3.4. Association between risk factors and bovine TB
Association of risk factors with PCR positivity was studied through bivariate analysis in each of the target population, and results are shown in Table 4. PCR positivity was the outcome, and the risk factors were treated as predictors. In Group A, contact with TB cases (OR = 23.5; 95% CI, 3.06–180.79) and raw milk consumption (OR = 6.34; 95% CI, 1.3161–30.5553) significantly increased the risk of positivity to nearly 20-fold and 6-fold, respectively (p < 0.05). A similar trend was also observed in Group C. Contact with active TB cases (OR = 3.7; 95% CI, 0.9612–14.4533) and raw milk consumption (OR = 5.3472; 95% CI, 1.9590–14.5956) significantly increased the likelihood of positivity by 3-fold and 5-fold, respectively. In Group C, exposure to animals (OR = 2.6308; 95% CI, 0.1297–53.3659) doubled the odds in favor of positivity; however, it was not statistically significant.
| Sr. No. | Risk factors | A | B | C | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Odds ratio (95% CI) | p | Z statistic | Odds ratio (95% CI) | p | Z statistic | Odds ratio (95% CI) | p | Z statistic | ||
| 1 | Previous contact with TB case | |||||||||
| No | 1 | 0.0024 | 3.034 | 1 | 0.8997 | 0.126 | 1 | 0.0571 | 1.903 | |
| Yes | 23.5 (3.0568–180.7912) | 1.2 (0.0540–27.6783) | 3.7 (0.9612–14.4533) | |||||||
| 2 | Living in a high endemic area | |||||||||
| No | 1 | 0.3415 | 0.951 | 1 | 0.2683 | 1.107 | 1 | 0.8914 | 0.137 | |
| Yes | 0.1472 (0.0028–7.6335) | 4.28 (0.3296–54.0878) | 0.8941 (0.1793–4.4584) | |||||||
| 3 | Animal contact | |||||||||
| No | 1 | 0.0187 | 2.352 | 1 | 0.5288 | 0.63 | 1 | 0.0502 | 1.959 | |
| Yes | 0.2237 (0.0642–0.7790) | 2.6308 (0.1297–53.3659) | 2.6496 (0.9993–7.0251) | |||||||
| 4 | Raw milk consumption | |||||||||
| No | 1 | 0.0213 | 2.302 | 1 | 0.7294 | 0.346 | 1 | 0.0011 | 3.273 | |
| Yes | 6.3415 (1.3161–30.5553) | 1.7556 (0.0723–42.6042) | 5.3472 (1.9590–14.5956) | |||||||
CI = confidence interval; PCR = polymerase chain reaction.
Represents association of PCR positivity (%) with risk factors.
4. Discussion
In industrialized countries, animal TB control and elimination programs, together with milk pasteurization, have drastically reduced the incidence of disease caused by M. bovis in both cattle and humans [12]. In developing countries, however, animal TB is widely distributed, control measures are not applied or are applied sporadically, and pasteurization is rarely practiced [13,14]. In India, very limited data on the zoonotic aspects of M. bovis are available. There is a lack of surveys to date to assess the public health concerns posed by bovine TB.
In the present study, we examined the incidence of bovine TB in different populations of Central India and also assessed the risk factors that may influence the occurrence of the disease. In view of the described objective, we primarily recruited participants who were in direct or indirect contact with animals.
The duplex PCR analysis conducted in our study indicated that the residents of the high TB endemic region of Central India had the highest number of M. bovis-infected cases (12.6%). This particular population was characterized by individuals living in a community with high crowding index and high prevalence of TB [15]. Majority of the population in this region consumed meat bought from local abattoirs. Some of the respondents during recruitment noted that no proper inspection of meat and meat products was done prior to consumption. Thus, the existing eating culture (eating of raw meat and drinking of raw milk) and the prevailing low standard of hygiene practices may be potential risk factors that favor the spread of bovine TB in this population. A report by Hubbert and Hagstad [16] suggested that ingestion of meat and meat products from slaughtered infected cattle that have not been thoroughly cooked may pose a serious risk of zoonotic infection.
Another important factor that may be contributing to the spread of zoonotic infection in this population was contact with TB index cases. The participants from this group lived in houses that were poorly ventilated and were household contacts of active pulmonary TB cases. In this study, we observed that the highest number of participants with clinical symptoms consistent with active TB such as fever, cough with expectoration, night sweats, chest pain, and abdominal pain, belonged to this group. It has been reported that M. bovis causes clinical presentations identical to those of M. tuberculosis-induced TB [8,14]. Also, most culture-based systems cannot differentiate between the two pathogens to the species level [17]. It may be likely that some cases that were earlier diagnosed with TB in our previous studies may in fact be overlooked cases of bovine TB.
The present study has significant clinical relevance because M. bovis is intrinsically resistant to an important first line anti-TB drug, i.e., pyrazinamide, and the standard regimen including this drug have to be altered [14]. Therefore, such studies would significantly help in patient management programs.
Subsequent high positivity was observed among participants belonging to Group A, consisting of farmers, dairy workers, and livestock keepers from rural settings. These individuals not only had close contact with the animals while feeding and milking, but also lived in close proximity at night, sometimes even under the same roof. A prospective cohort study among farmers in the United Kingdom suggested that agricultural workers may acquire bovine TB by inhaling cough spray from infected cattle and develop typical pulmonary TB [18]. According to one of the respondents, they share their room with the newly born calves at night to protect them from wild animals. Consumption of raw milk was also a common practice in this group. During the interviews, it was recorded that many participants preferred unpasteurized milk over boiled milk because of its richer taste. This could be one of the reasons for transmission of M. bovis from cattle to humans [19]. It has also been postulated by Michel et al. [20] that pastoralist and rural communities would be at greatest risk for bovine TB, but the lack of data for these population groups prevents confirmation of this assumption.
The third population recruited for the study consisted of zookeepers and animal handlers. The possible mechanisms of transmission of M. bovis in this particular population may include close contact while handling, cleaning the barn, participating in animal necropsies, and living in close proximity to the cages. A report by Michalak et al. [21] described the first case of reverse zoonosis wherein M. tuberculosis was transmitted from humans to elephants. Therefore, reliable diagnosis and prevention of TB in all domesticated and exhibited animals is ideal.
To the best of our knowledge, this is the first report on the prevalence of bovine TB in the Central Indian population. A systematic literature search on the occurrence of zoonotic TB by Müller et al. [22] showed that there is lack of data for the World Health Organization region of Southeast Asia, including major cattle producing middle- and low-income countries (e.g., India, Bangladesh, Pakistan, Myanmar, and Indonesia). Recorded incidence rates for zoonotic TB in Europe, the United States, Australia, and New Zealand were consistently below 1/100,000 population/y. The incidence rates were not available for other countries [22].
Individual studies from various regions reported high proportions of zoonotic TB for specific population groups and settings. For example, in the Hispanic community in the United States, zoonotic TB appeared to be a considerable proportion of all TB cases and was associated with the consumption of unpasteurized cheese from Mexico. The highest median proportions for TB caused by M. bovis were observed in countries in Africa: Ethiopia, Nigeria, and Tanzania. However, the specific populations affected and risk factors of zoonotic TB in these settings remained largely elusive [14]. In keeping with the earlier reports, our present study also indicates pockets of zoonotic transmission of TB for specific population groups and settings.
In the Indian context, studies by Shah et al. [17] and Prasad et al. [23] have shown high incidence of M. bovis and M. tuberculosis in extrapulmonary samples of humans and cattle, respectively. A similar study by Mittal et al. [24] has demonstrated the importance of screening and differential diagnosis of MTBC in humans and livestock.
Despite being an insightful study, our work suffers from the limitation of insufficient data on animal health. It was learned that worshipping of cattle has spiritual significance in some regions; therefore, withdrawing blood or performing any tests on their animals was not possible. The present study thus needs further evaluation in animal population to identify the main transmission drivers in these areas.
5. Conclusion
In conclusion, this study has documented the prevalence of neglected bovine TB in human population in Central India. Diagnosis and monitoring of this disease are essential, especially in developing countries such as India, where humans and animals have close association in routine life. This study would thus be valuable in guiding policy makers for further studies in bovine TB epidemiology.
Abbreviations
- TB
Tuberculosis;
- M. bovis
Mycobacterium bovis;
- MTB
Mycobacterium tuberculosis;
- MTBC
Mycobacterium tuberculosis complex species;
- RD
Region of difference;
- PCR
Polymerase chain reaction;
- AFB
Acid fast bacilli;
- CIIMS
Central India Institute of Medical Sciences;
- PBS
Phosphate buffered saline;
- EDTA
Ethylenediaminetetraacetic acid;
- CTAB
Cetyl-trimethyl-ammonium bromide;
- BCG
Bacille Calmette Guerin;
- ESAT-6
6 kDa Early Secretory Antigenic Target.
Conflicts of interest
None.
Acknowledgments
This work was supported by the Indian Council of Medical Research (ICMR), New Delhi, India (Grant No. Zon.15/11/2014-ECD-II). The funding source had no involvement in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the article for publication. The authors acknowledge Dr Dewanand R. Kalorey, Dr Nitin V. Kurkure, and Dr Sandeep P. Chaudhari for their support. The authors also thank Ms Sonali Manke and Ms Ankita Tiwari for their technical assistance.
Appendix A. Supplementary data
Supplementary data associated with this article can be found, in the online version, at http://dx.doi.org/10.1016/j.jegh.2017.08.007.
References
Cite this article
TY - JOUR AU - Prachi R. Bapat AU - Renuka S. Dodkey AU - Seema D. Shekhawat AU - Aliabbas A. Husain AU - Amit R. Nayak AU - Anuja P. Kawle AU - Hatim F. Daginawala AU - Lokendra K. Singh AU - Rajpal S. Kashyap PY - 2017 DA - 2017/09/13 TI - Prevalence of zoonotic tuberculosis and associated risk factors in Central Indian populations JO - Journal of Epidemiology and Global Health SP - 277 EP - 283 VL - 7 IS - 4 SN - 2210-6014 UR - https://doi.org/10.1016/j.jegh.2017.08.007 DO - 10.1016/j.jegh.2017.08.007 ID - Bapat2017 ER -
