Noninvasive evaluation of large artery function and structure in dialysis patients: Clinical applications
- DOI
- 10.1016/j.artres.2010.10.032How to use a DOI?
- Keywords
- Calcification; Pulse wave velocity; Stiffness; Augmentation index; Dialysis
- Copyright
- © 2010 Published by Elsevier B.V. on behalf of Association for Research into Arterial Structure and Physiology.
- Open Access
- This is an open access article distributed under the CC BY-NC license.
Introduction
Cardiovascular disease (CVD) remains a major cause of morbidity and mortality in patients with chronic kidney disease (CKD), either treated by hemodialysis, peritoneal dialysis or after renal transplantation, as well as in earlier stages of CKD not yet requiring renal replacement therapy. Several functional and structural arterial alterations can be detected before the appearance of clinical manifestations, including endothelial dysfunction, changes in arterial geometry and biomaterial properties of the vascular wall, increased reflective properties of the vascular system, and vascular calcification. Although highly prevalent among patients with CKD, these abnormalities are not specific and also occur in other populations at high risk for incident CVD and with ageing. This has led to the concept of renal disease as a model of accelerated vascular ageing where vascular changes can already be observed at young age. The clustering of traditional risk factors in patients with CKD only partially explains the high incidence of CVD, and after multivariate adjustment, kidney function expressed as estimated glomerular filtration rate, remains an independent predictor of outcome.1 Therefore research has focused on factors specific to the uremic condition leading to the identification of an increasing number of uremic retention solutes with direct vasculotoxic effects.2 The complexity of the different biochemical pathways involved, superimposed on the heterogeneous nature of the CKD population (underlying kidney disease, rapid changes in dialysis practice and post-transplant care, etc.), has stimulated the interest in markers of subclinical vascular damage. Such markers should represent at least intermediate endpoints of clinical outcome and ideally be surrogate endpoints with a proven relationship between improvement of the parameter and a reduction in events. Moreover, techniques for their assessment should be accurate, reproducible and noninvasive. In the renal population, several established surrogate endpoints such as left ventricular hypertrophy or albuminuria are often difficult to interpret, non-specific, or even unavailable, depending on the underlying kidney disease, residual kidney function and/or dialysis modality. Therefore, other noninvasive methods have been used for assessing acute hemodynamic changes as well as long term functional and structural alterations of large arteries, in different subpopulations of CKD.
Acute hemodynamic effects of dialysis
Acute hemodynamic changes, mostly episodes of hypotension, during hemodialysis (HD) are well known to be related to worse outcome. Most physicians presume that an acute peritoneal dialysis (PD) dwell does not produce acute hemodynamic changes, which might explain why the topic has been investigated only to a limited extent. The few studies evaluating changes in hemodynamic parameters indicate however that some mild changes in blood pressure (BP) and vascular tone do occur during an acute dwell.3,4 Such repetitive undulations in hemodynamic status can potentially be harmful, as they might increase cardiac workload, induce oxidative stress and can negatively impact residual renal function.5 The intraperitoneal (IP) instillation of dialysate causes an abrupt increase in the IP pressure. This could enhance venous retour to the right ventricle and this way improve cardiac output, or reduce venous return by compressing the vena cava inferior and decrease cardiac output.
Ivarsen et al.6 analyzed the evolution of cardiac output using foreign gas rebreathing in 15 PD patients, following the instillation of 2–3 L of PD fluid, and this in the supine and upright position. In the supine position, no effect of a 2 L infusion was observed. In the upright position, a decrease in cardiac output, and an increase in peripheral systemic resistance was observed, resulting in an absence of a net change in BP. Remarkably, the results were not different when either using a 2- or a 3-L fill, and no change in heart rate was observed. Boon et al.,3 using fingertip continuous pressure monitoring, found an increase in BP and peripheral resistance after instillation of 2 L of glucose based peritoneal dialysate, an effect they attributed either to compression of the mesenteric resistance vessels or to the low temperature. Unfortunately, in these studies, hypertonic glucose solutions were used, so an eventual impact of hyperosmolarity and/or glucose cannot be excluded. By instillation of a non-glucose containing solution (icodextrin; Baxter Healthcare, Ireland), at neutral pH and warmed to 37.0°C, we demonstrated that the increase in IP pressure (2.3cm H2O) closely paralleled a change in hemodynamic parameters,7 pointing to the impact of IP volume per se. The effect of IP volume occurs mostly in the beginning of the filling procedure, with a rise in carotid systolic BP and augmentation pressure of 4.7 mmHg and 3.1 mmHg, after infusion of the starting volume. Augmentation index (AIx) increased by 5.7% and heart rate decreased by 2.6/minute, while carotid-femoral pulse wave velocity (PWV) remained unchanged. No additional hemodynamic changes except for a rise in diastolic BP with increasing volume were observed after subsequent addition or removal of volumes. These effects persisted until complete drainage of the abdomen and may be due to an enhanced preload, resulting from IP venous compression, and/or increased wave reflection. Of note, changes in brachial systolic and diastolic BP were not significant, illustrating the importance of studying central hemodynamic parameters based on accurately calibrated tonometric waves.8 A rise of 4.7 mmHg in central systolic pressure may appear to be small, but is similar to the difference in the two treatment arms of the CAFÉ study (4.3 mmHg), considered to be responsible for the different clinical outcomes.9 The observed hemodynamic effects are in line with a study by Selby et al.10 where in automated peritoneal dialysis patients, BP decreased during the drain, and increased during the instillation of dialysate (unfortunately all glucose based). The decrease in BP was correlated with a decrease in peripheral resistance and an incomplete compensatory rise in cardiac output.
Using hypertonic 3.86% glucose, Selby et al.4 found an increase in BP and cardiac output during the dwell, irrespective of the biocompatibility of the solution used. Our group,11 in contrast, was not able to confirm these results. Despite a significant rise in serum glucose and insulin levels over time, more pronounced during the high glucose dwell, there was no demonstrable effect on central systolic or diastolic BP in this study. One of the reasons for the discrepancies between these two studies may relate to the different methodology used to measure hemodynamic parameters. We chose to set up this study using the SphygmoCor system (AtCor Medical, Sydney, Australia) and to measure at the level of the carotid artery, as this approach yields more accurate estimates of central hemodynamic measurements.12 Finger arterial pressure tracing with Modelflow pulse wave analysis (Finometer; Finapres Medical Systems, Arnhem, The Netherlands) as used in studies by Selby,4,10,13 in contrast, has only been validated for assessing brachial artery pressure14 and has been shown to be insufficiently accurate in specific conditions, such as pregnancy.15
Long term effects of anemia and Vitamin D deficiency
Physiologic laminar fluid shear stress (SS) is responsible for endothelial cell survival and quiescence and their secretion of substances that favour anticoagulation, inhibit inflammation, induce vasodilation, and exert atheroprotective effects in vivo.16,17 In patients with end-stage renal disease, outward remodeling, arterial stiffness, and impaired endothelial function are observed. Flow-mediated dilation is often used as a reference method for studying biomarkers of endothelial dysfunction or evaluating the effect of interventions. In clinical studies, non-pharmacalogical interventions used to increase flow are distal limb ischemia or skin heating. Although the resulting vasodilatatory reponse is termed flow-mediated dilation (FMD), the actual physical stimulus acting upon the endothelial surface is the increase in shear stress (SS), not the increase in flow itself. Endothelial dysfunction and arterial stiffness appear to be linked, although the exact mechanisms remain unclear. High SS induces acute changes of the conduit artery function (FMD)18 and arterial remodeling with modified arterial wall properties.19 The prognostic importance of an impaired FMD was demonstrated in a study of 78 HD patients, showing that lower postischemic forearm reactive hyperemia was predictive of all-cause mortality, independent of aortic PWV.20
With the advancement in electronics and computer technology, it became possible to directly measure the shear rate (SR) close to the arterial wall (endothelial surface), which enables, in combination with the measurement of whole blood viscosity (WBV), to quantify SS as the product of SR × WBV. With a high-resolution echo-tracking device (Wall-track System; Pie Medical, Maastricht, The Netherlands), changes in flow velocity profiles and diameter were studied in 44 HD patients, following progressive hand warming-induced hyperemia. The use of skin heating as flow stimulus allows to quantify the vasodilation in response to different known levels of endothelial stimulation. Using this approach, the flow-independent negative impact of anemia-associated low WBV on levels of SS could be demonstrated.21 The concurrent improvement in SS and FMD following anemia correction by erythropoietin administration supports a causal role of anemia in endothelial dysfunction, consistent with the known adverse effect of low SS on endothelial function.22 Because SS is the real physical stimulus acting upon the endothelium, future studies should take into account the independent effects of both flow and blood viscosity. Likewise, the results of interventions such as erythropoietin treatment looking only to hemoglobin/hematocrit (as a proxy for WBV) may be difficult to interpret without any information on arterial wall and blood flow characteristics. A benefit can only be expected when there is at least some degree of endothelial function left and when the artery is capable of dilating at all (preserved endothelial independent vasodilation), which may not be the case in the majority of patients included in previous randomized trials. This may be one explanation for the seemingly contradictory results of our and previous studies23–25 compared to those of intervention studies showing no benefit or even a worse outcome at higher levels of target hemoglobin.26–28 Moreover, in our study, care was taken not to “overcorrect” (i.e. normalize) the anemia to levels as achieved in the higher hemoglobin/hematocrit arms of these randomized trials.26–28
The role of 25(OH) Vitamin D and 1–25(OH)2 Vitamin D in the pathophysiology of Chronic Kidney Disease – Mineral and Bone Disorder (CKD-MBD), including CVD complications, continues to be an area of intensive research. Most large epidemiological studies and interventional trials focused on either PTH, calcium and phosphorus levels, or the use of active Vitamin D analogues, producing rather inconsistent or even conflicting results. Nutritional 25(OH)Vitamin D deficiency is highly prevalent in renal populations and supplementation is safe and inexpensive. In a cross-sectional study of 52 HD patients, we observed a consistent and independent relationship between a deficiency in both Vitamin D forms and increased arterial stiffness, reduced distensibility as well as endothelial dysfunction, pointing to a potential new mechanism involved in the functional and structural alterations of large arteries in dialysis patients.29 Since calcification status and abnormalities in serum levels of calcium, phosphorus or PTH, were not independently related to Vitamine D status, studies addressing CVD risk in relation to CKD-MBD should measure both vascular calcification as well as stiffness. Future studies should further establish the causal role of nutritional and hormonal vitamine D deficiency in inducing stiffness, and the pathophysiological pathways involved. This information, in turn, could also help to better define the indications for treatment with active Vitamine D or their analogues.
Calcification, stiffness and wave reflections in epidemiologic studies
The Calcification Outcome in Renal Disease (CORD) study is currently the largest study in dialysis patients to assess at the same time parameters of arterial stiffness, wave reflection and vascular calcification, and their predictive value for patient outcome. In this epidemiologic study, abdominal aortic calcification, carotid-femoral PWV, and central AIx were measured in 1084 dialysis (both HD and PD) patients, recruited from 47 European dialysis centers across 6 European countries. Calcification was assessed by semiquantitative scoring of a plain lateral lumbar radiograph (AAC scores) according to a validated method.30 In previous studies, methods used to quantify the extent of calcification were more expensive, like electron beam computed-tomography,31 or more time-consuming with the use of both utrasonographic and radiographic imaging at different anatomical sites.32 Although computed-tomography based methods remain the golden standard for quantification, a plain lateral lumbar X-ray is far less expensive and readily available. Moreover, a recent study demonstrated that abdominal X-ray scoring had a very good discriminatory value compared with coronary artery calcification (CAC) scores, with an area under curve of the receiver-operating characteristic above 0.70 at each CAC threshold.33
The cross-sectional evaluation of the abdominal aortic calcification clearly showed the magnitude of the problem nephrologists are facing.34 Over 80% of patients had visible calcifications (AAC score ≥ 1), and in more than half of the population all 4 lumbal levels were affected. The number of patients affected and the severity of calcification increased progressively from L1 to L4, indicating that calcification developed in a distal to proximal direction, consistent with findings of large post-mortem studies performed in the sixties.35
As in recent analyses of the population-based Rotterdam study36 and the Anglo-Cardiff Collaboration Trial,37 as well as in a study of 131 HD patients,38 PWV was independently related to abdominal aorta calcification,39 supporting the prevailing hypothesis that calcification in dialysis patients is predominantly medial wall calcification which contributes to stiffness. The progressive increase in PWV with increasing quintiles of AAC scores is illustrated in Fig. 1. In contrast, AIx only weakly correlated with AAC and was mainly related to heart rate and anthropometric factors. Although calcification and increased PWV clearly clustered in older patients with established CVD, still 1/3 of apparently low risk patients had aortic calcifications and/or abnormal values of PWV or AIx. Since these patients are generally considered candidates for renal transplantation, assessment of abdominal aortic calcification, PWV and AIx would be of particular value in this population. Detection of abdominal calcification in transplant candidates could also prompt further imaging of the adjacent iliac arteries and result in an early detection of potential lesions that may be relevant for the surgical procedure. The cross-sectional data of the CORD study also confirm the notion that CKD is a metabolic disorder characterised by accelerated vascular ageing. Indeed, CORD patients aged 25–35 years had a mean AIx and PWV comparable with values observed in the fifth decade in healthy persons (Fig. 2).
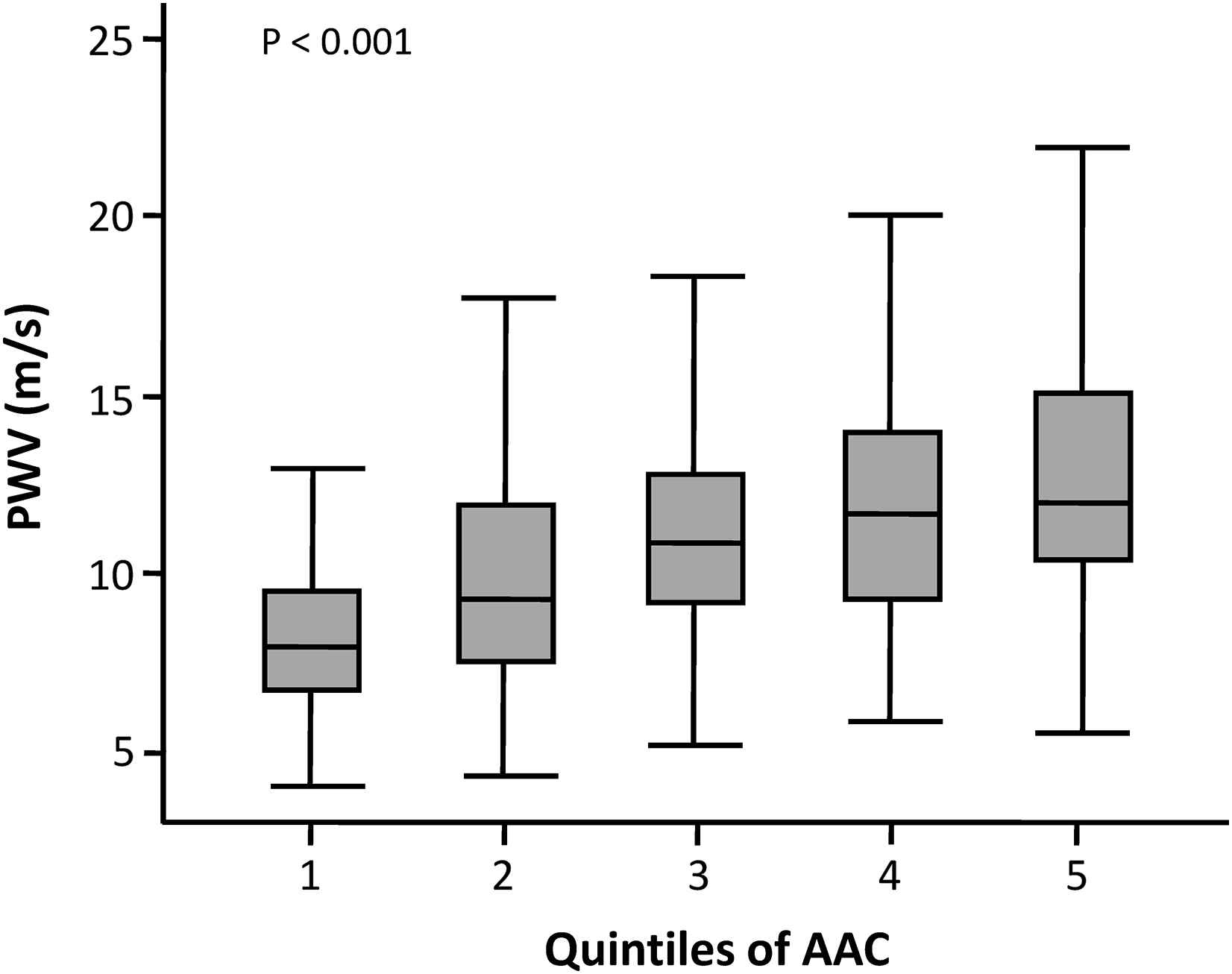
Relationship between AAC scores (quintiles) and PWV in the CORD study.The lowest quintile of AAC scores corresponds to the subgroup of patients without any visible calcification (AAC=0).
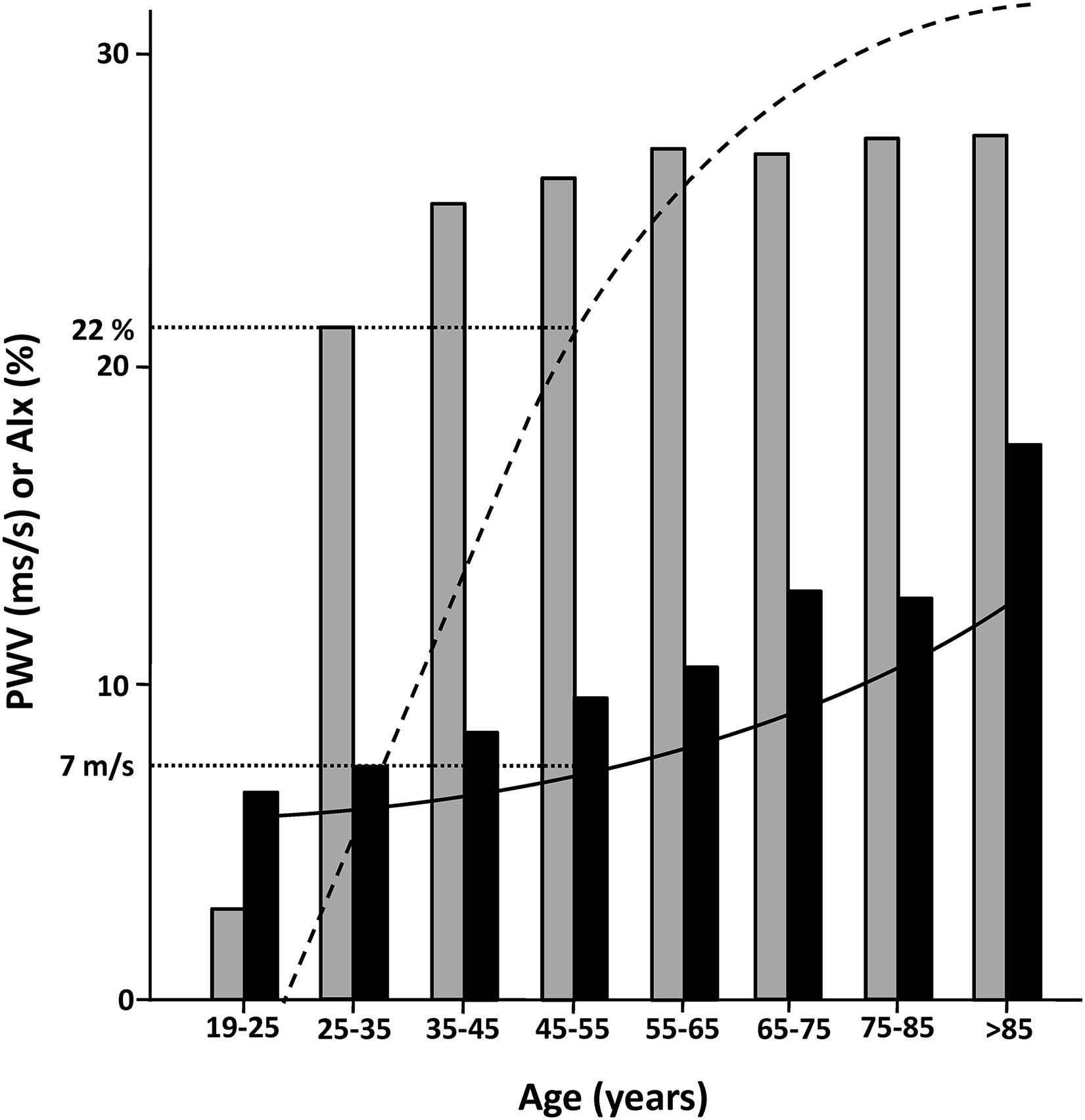
Mean AIx (grey bars) and PWV (black bars) according to age in CORD patients compared with the general population (adapted from McEniery et al. J Am Coll Cardiol 2005;46:1753--60; dashed and solid lines represent average AIx and PWV, respectively). Values in CORD patients at age 25–35 years are comparable with those of the general population at age 45–55 years (dotted lines).
In the CORD study, after a median follow-up of 2 years, 234 deaths and 91 nonfatal cardiovascular events were recorded, corresponding to an incidence rate of 15% per year. AAC and PWV each individually were strong predictors of outcome, confirming earlier studies in HD patients. The combined information of AAC and PWV, however, further improved risk prediction, despite the fact that abdominal aortic calcification and PWV are strongly related.40 This provided evidence that both should be measured when assessing CVD risk in the dialysis patient.
Additional analyses of the CORD data are in progress, and some preliminary results have been reported in abstract form. It was shown that the change in PWV over the two year observation period was related to outcome, independently from the baseline value, which further supports the role of PWV as a surrogate endpoint and its potential value for the follow-up of patients [Verbeke F et al., J Am Soc Nephrol. 2007; 18: 69A (FC006)]. In a similar analysis of the change in AAC scores over time, it was found that calcification progressed by 2 AAC score points per year [Honkanen E et al., J Am Soc Nephrol. 2008; 19: 718A (PO2693)] as compared to 0.3 points in the historical Framingham cohort.30 Finally, from an analysis of different parameters of wave reflection, central augmentation pressure was superior in predicting outcome compared to peripheral and central augmentation index [Verbeke F et al. J Am Soc Nephrol. 2008; 19: 11A (FC047)].
Conclusions and future perspectives
With the use of applanation tonometry, it was demonstrated that an acute instillation of PD fluid increases central but not peripheral BP. The effects are attributed to the increase in IP pressure during the filling procedure and persist for several hours. Although the underlying mechanisms remain incompletely understood, the concommitant rise in AIx suggests that enhanced wave reflection may play a role. The effect of hyperglycemia is debated, but the use of nonhypertonic glucose based solutions seem to be without or of very limited hemodynamic effect. Therefore, from the available PD regimens, those with high volume, fast cycling hypertonic exchanges should be avoided.
The evaluation of endothelial function by FMD is time-consuming, requires a high level of technical expertise as well as an expensive ultrasound equipment, but provides the most complete and detailed information. It allowed to demonstrate that correction of anemia by erythropoietin administration improves FMD by increasing SS, an effect that is attributable, at least in part, to an increased WBV. This information is clinically relevant because it had been shown previously that impaired endothelial function is predictive of mortality. Another advantage is that endothelial function can be affected on the short term whereas changes in structural properties of the arterial wall, such as stiffness and calcification, may only occur over long periods of time.
CORD is the first study that implemented simple and inexpensive yet reliable and validated methodologies on a large scale and in daily clinical conditions. CORD has paved the way for the use of AAC-scoring and applanation tonometry in daily clinical nephrology practice and in future studies. Meanwhile, PWV has been adopted as a marker of target organ damage in the latest 2007 ESH/ESC hypertension guidelines, and the accumulating evidence supporting the independent prognostic value of vascular calcification urges to consider also this parameter for CVD risk assessment in renal populations. Following the logic of disease prevention, the next step will be to look for therapeutic strategies that may affect abdominal aortic calcification and PWV favourably and hopefully will also improve outcome. Obviously, the maximal benefit is to be expected from screening and intervention at earlier stages of kidney disease. Therefore, efforts should focused on patients with CKD stage 3–5 not yet on dialysis, or even earlier like in patients with heavy proteinuria and at high risk for rapid loss of kidney function, or in persons that are considered for living kidney donation.
The use of these measures will be valuable in the design and interpretation of future randomized trials and help us to understand the negative or conflicting results of previous studies. It should be verified whether treatment with calcium-containing versus calcium-free phosphate binders in patients with different levels of abdominal aortic calcification and PWV at the start affects these parameters and subsequent outcomes in the same way. The consistent cross-sectional relationship between nutritional Vitamin D deficiency and indices of arterial stiffness and endothelial dysfunction, points to one of the therapeutic targets to be prioritized in future studies and may be part of a pathophysiological link between bone and mineral disorders, and CVD in dialysis patients. Such interventional studies will also help to define therapeutic thresholds as well as limits of abdominal aortic calcification and PWV values beyond which an intervention is unlikely to affect outcome
Conflicts of interest/disclosures
F Verbeke, W Van Biesen, and R Vanholder have acted as study investigators for the CORD study, funded by Genzyme. F Verbeke has served as consultant to Genzyme.
References
Cite this article
TY - JOUR AU - Francis Verbeke AU - Wim Van Biesen AU - Raymond Vanholder AU - Luc Van Bortel PY - 2010 DA - 2010/10/27 TI - Noninvasive evaluation of large artery function and structure in dialysis patients: Clinical applications JO - Artery Research SP - 138 EP - 143 VL - 4 IS - 4 SN - 1876-4401 UR - https://doi.org/10.1016/j.artres.2010.10.032 DO - 10.1016/j.artres.2010.10.032 ID - Verbeke2010 ER -
